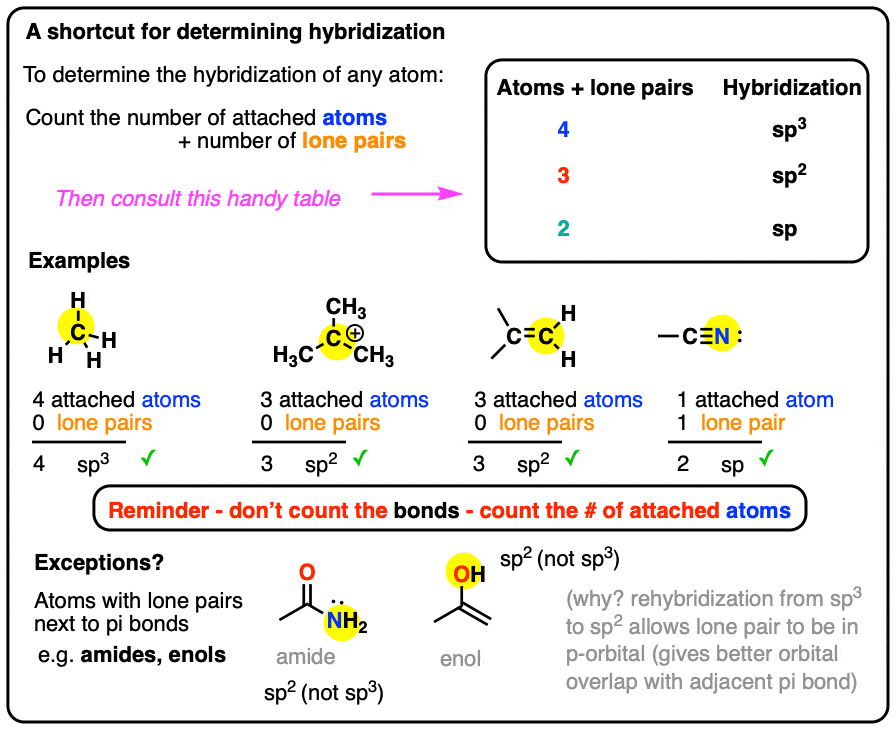
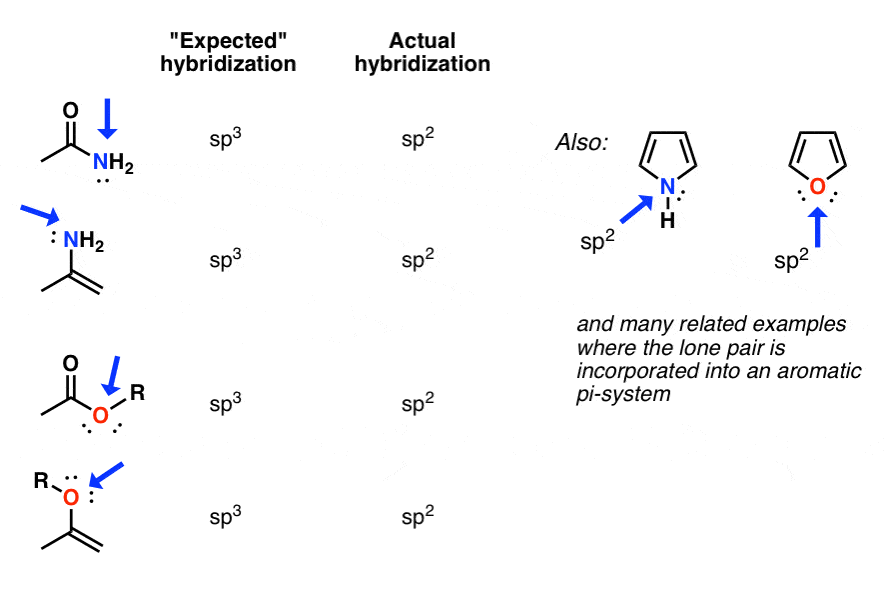
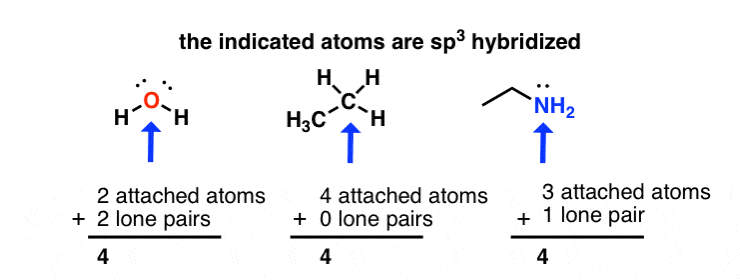
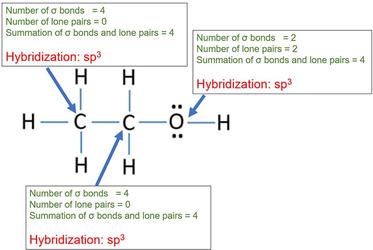
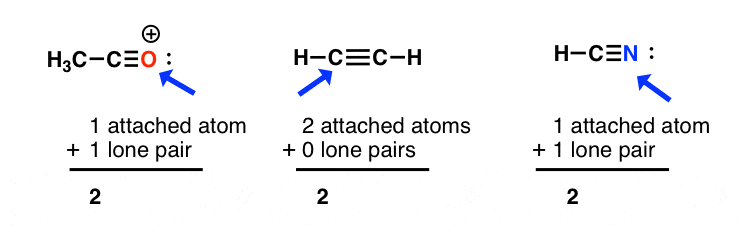
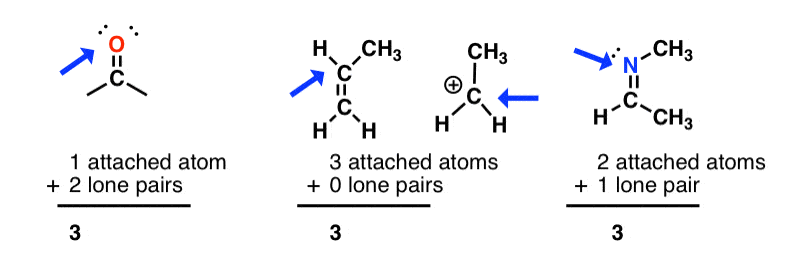
How to Determine Molecular Geometry Based on the Steric Number & Hybridized Orbitals | Chemistry | Study.com

Assertion: Ammonia shows a trigonal pyramidal molecular structure.Reason: In the structure of ammonia, three atoms are attached to the central atom and thus, shows tetrahedral electron pair geometry.
_how-to-determine-hybridization-s-sp-sp2-and-sp3-organic-chemistry.jpg)
Hybridisation Hybridization (sp, sp2, sp3) Chemical Bonding A level H2 JC Chemistry Tuition Centre from r sxdxnognk Watch Video - HiFiMov.co
✓ Solved: Give the expected hybridization of the central atom for the molecules or ions from Chapter...

hybridisation of SF4 in this formula Hybridization=1/2(valency electron in central atom+no. Of atom attached - Brainly.in

Determining Hybridization of Valence Orbitals in Covalent Molecules using VSEPR Theory | Chemistry | Study.com