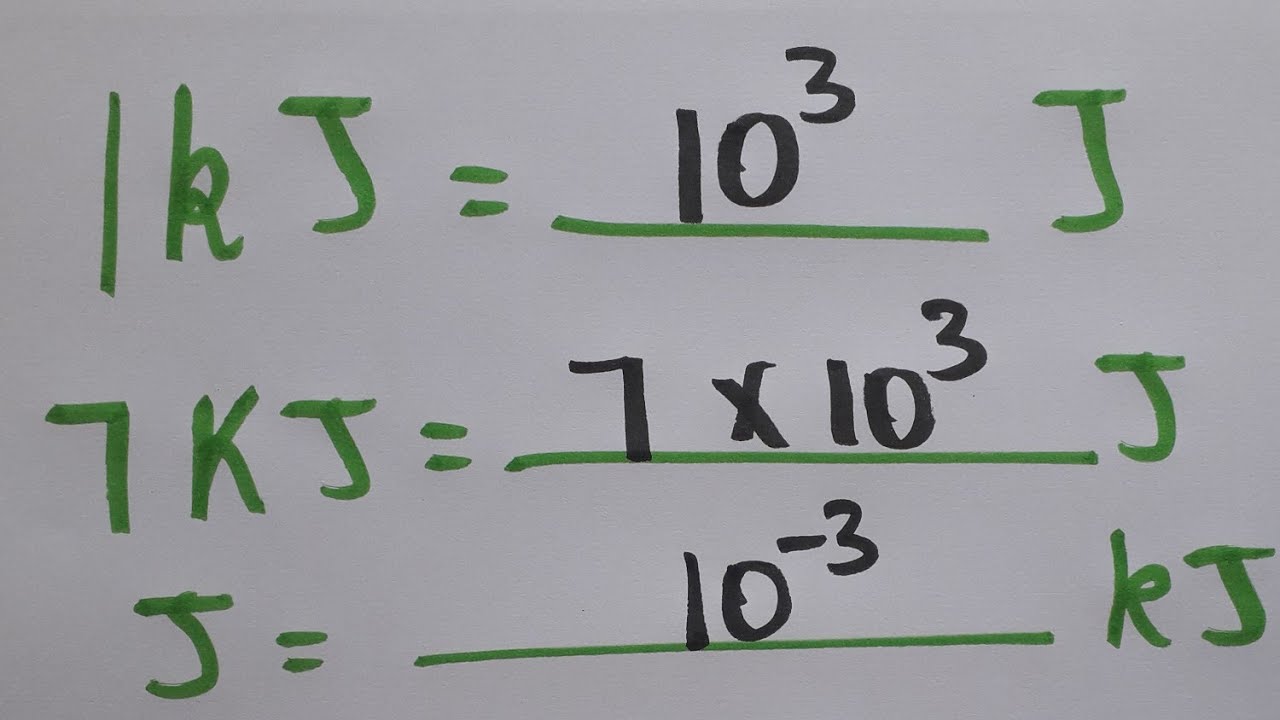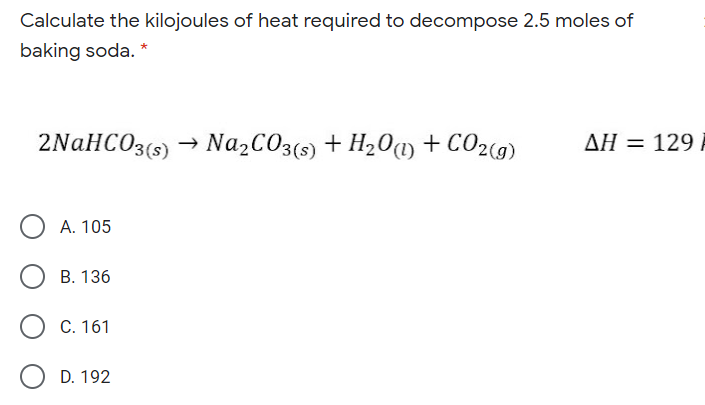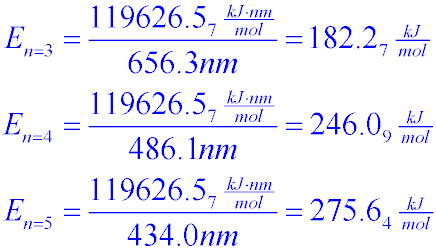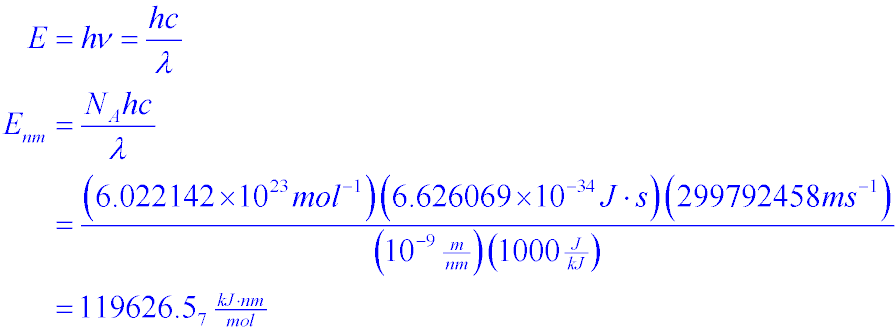Given:N2 + 3H2 → 2NH3Bond Bond Energy (kJ/mol)N≡N 942H–H 432N–H 386Use the bond energies to calculate - Brainly.com

Calculate the number of KJ of heat necessary to raise the temperature of 60.0g of aluminium from.... - YouTube

Question Video: Conversion between Kilocalories and Kilojoules Using the Energy Consumption of the Adult Human Body | Nagwa

SOLVED: Calculate the fuel value of coal in kilojoules per gram. The heat capacity of the bomb calorimeter is 9.96 kJ/°C fuel value= kJ/g 1 gram= 23.5 C 2 gram= 26.9 C 3 gram= 30.4 C

Calculate the energy change (in kilojoules per mole) for the formation of the following substance from their elements. LiF (The sublimation energy for Li(s) is +159.4 kJ/mol, the | Homework.Study.com